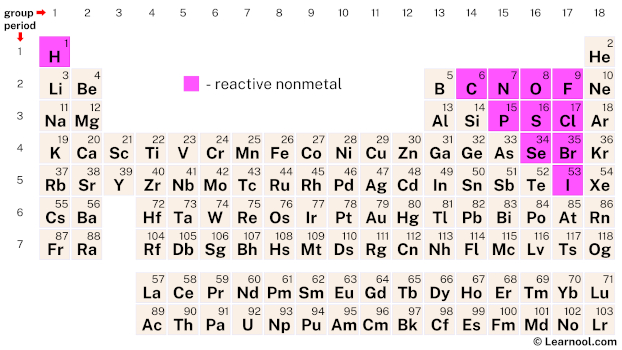
 Metal reactivity relates to ability to lose electrons. The trend in reactivity in the nonmetals is the opposite of the trend in the metals. Four major factors affect reactivity of metals: nuclear charge, atomic radius, shielding effect and sublevel arrangement (of electrons). The most reactive nonmetal is fluorine, F. 
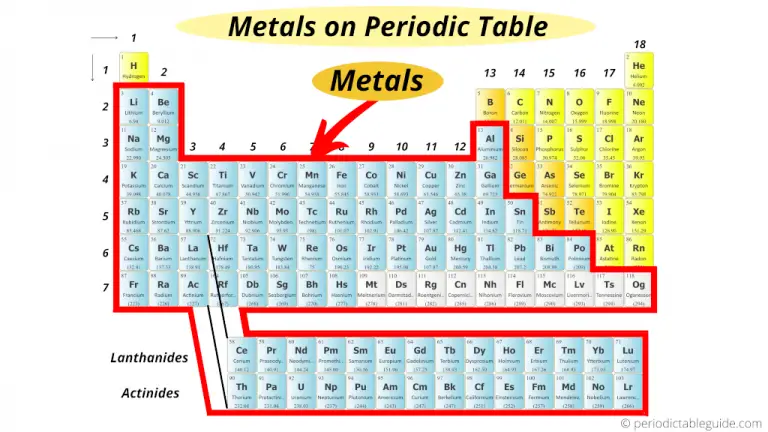
Nonmetals are separated from metals by a line that cuts diagonally through the region of the periodic table containing elements with partially filled p orbitals. The elements within the same group of the periodic table tend to exhibit similar physical and chemical properties. The nonmetals are located on the upper right side of the periodic table. Nonmetals include solids, liquids, and gases at room temperature and pressure. 
They tend not to be malleable or ductile, so they form brittle solids.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
